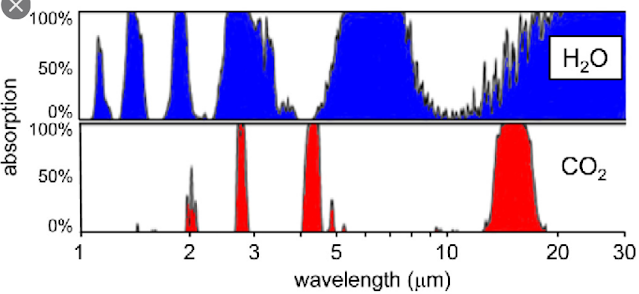
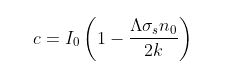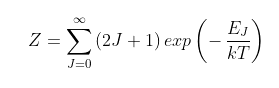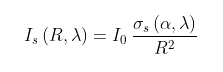In my previous three posts I have explained how the Greenhouse Effect (GHE) works on Earth, and how it is affected by changes to the carbon dioxide (CO2) concentration in the atmosphere. The problem with studying the GHE on Earth, though, is that its operation is complicated by the presence of large amounts of water vapour in the atmosphere. As water vapour also has a broader absorption band and higher atmospheric concentration than CO2, this means that changes in the CO2 concentration are less important than they would be otherwise. If we want to understand and measure the GHE just due to carbon dioxide, then we need an environment with high levels of CO2 but low levels of other greenhouse gases. In this respect one of the best places to study is Mars.
The Atmosphere of Mars
The Martian atmosphere has some similarities with Earth but many differences. It contains many of the same gases (nitrogen, oxygen, water vapour, argon, CO2), but the proportions are vastly different. The atmosphere of Mars is 96% CO2 with about 2% nitrogen and 2% Argon (although different pages on Wikipedia give slightly different values such as 95% CO2 and 3% nitrogen). There are also trace levels (< 0.1%) of other gases such as water vapour (210 ppm) and oxygen (0.15%).
The other main difference in terms of the atmosphere is the pressure at the surface. At 610 Pa this is only 0.60% of the surface pressure on Earth, and as about 96% of this is CO2, this means that there is a surface density of 3610 mol/m2 of CO2 on Mars compared to only 150 mol/m2 on Earth. So any outgoing radiation from the surface of Mars has 24.06 times as much carbon dioxide gas to penetrate, in order to escape into outer space, compared to on Earth. What this means in practice is that the Greenhouse Effect in the Martian atmosphere should be easier to analyse because it can only have one source - CO2.
The Energy Balance for Mars
Mars is also approximately 52% further from the Sun than is Earth, so one might expect that to mean that its surface is colder. This is true, but not as much as it should be based on distance alone.
The solar radiation flux entering the Martian atmosphere is only 586 W/m2 compared to the 1360 W/m2 that irradiates the Earth. As this radiation is spread over a surface area (4πr2) that is four times greater than the cross-sectional area of the planet (πr2) in each case, this means that the average radiation flux at the Martian surface is 143.5 W/m2. Yet this is only 22% less than the 184 W/m2 that reaches the Earth's surface. This is because nearly 50% of incident radiation on Earth is either reflected by clouds or is absorbed by ozone and water vapour in the upper atmosphere. But there are no clouds, ozone or significant water vapour on Mars.
Then there is the issue of surface albedo or Bond albedo. This is the proportion of incident radiation that is reflected by the planet back out into space without being absorbed, either from clouds or the surface. For Earth this is about 31%; for Mars it is only 25%. This means that the average surface absorption on Mars is 108 W/m2 compared to 161 W/m2 on Earth. So while Mars only receives 43% of the solar radiation that Earth does, after absorption and reflection the Martian surface receives 67% of the radiation that the Earth's surface does. That is a relative increase of more than 50% for Mars which partially compensates for its greater distance from the Sun.
Calculating the Surface Temperature of Mars
If we now invoke the Stefan-Boltzmann law (see Eq. 13.1 in Post 13),
I = σT4
(89.1)
where I is the surface radiation flux, σ is the Stefan-Boltzmann constant, and T is the surface temperature in kelvins, we see that a surface radiation flux of 108 W/m2 equates to a mean surface temperature on Mars of 209 K (or -64°C). Yet the true mean temperature is thought to be about 215 K (or -58°C). The difference is due to the Greenhouse Effect.
For comparison, on Earth a solar flux at the surface of 161 W/m2 would equate to a mean surface temperature of 231 K (or -42°C), yet the true mean temperature is about 289 K (or +16°C). So the GHE on Earth adds 58°C of warming while on Mars it only adds 6°C. Yet there is almost twenty-five times more carbon dioxide on Mars (3690 mol/m2) than on Earth (150 mol/m2), so you would expect the greenhouse effect due to CO2 to be stronger. But how much stronger? The answer is: not very. In fact it is significantly less than the actual measured value on Earth.
Calculating the Strength of the GHE on Mars
In Post 87 I showed how the width of the CO2 absorption band at 15 µm can be determined using the known concentration of CO2 (No), its scattering or absorption cross-section (σs), the quantized frequency of rotation of the CO2 molecules (B), and the temperature (T). From this it is possible to estimate the critical CO2 concentration needed to absorb most of the infra-red radiation (Nth). The term Nth can be estimated to be 0.5 mol/m2 based on the value of σs, while No will be about half the total CO2 concentration, so No = 1845 mol/m2. From this we can estimate the maximum number of excited rotational states in the absorption band, Jth, using (see Post 87)
where k is Boltzmann's constant, h is Planck's constant and Z is a normalization constant (see Eq. 87.2 in Post 87) equal to 185.5 in this case. The ratio term kT/hB has the value 185.3 (as hB = 0.1 meV and T = 215 K) and is always approximately equal to the Z value.
The result we obtain is that Jth = 36.3, which, given that the spacing of the bands is 0.2 meV, means that the absorption band has a width of 14.52 meV and extends from a wavelength of 13.78 µm to 16.44 µm. This compares to a calculated range of 14.00 µm to 16.14 µm for the same band on Earth, although the measured width on Earth is actually found to be from about 13.35 µm to 17.35 µm. So even though the calculated width of the 15 µm absorption band for Mars is slightly larger than the equivalent for Earth, it is not significantly greater. But it is significantly less than the measured band width on Earth.
The impact of the high Martian atmospheric CO2 concentration on the radiation feedback is demonstrated in Fig. 89.1 above. The red curve indicates the proportion of the outgoing infra-red radiation (blue curve) that is reflected by the CO2 molecules and it amounts to f = 12.5%. This is slightly more than the 10% seen for the GHE due to backscattering from CO2 on Earth (see Post 87) despite the absorption band being further from the peak in the emission spectrum due to the lower surface temperature on Mars.
Calculating the Temperature Rise
The radiation feedback of f = 12.5% shown in Fig. 89.1 equates to a 14.3% increase in the surface radiation and also of T4 (because of Eq. 89.1), which then equates to a 3.4% increase in T. So if the initial surface temperature on Mars without the GHE was 209 K, the temperature with the GHE included will be 3.4% greater, or 216.1 K. This means that the temperature rise at the surface due to the Greenhouse Effect is expected to be 7.1 K, which is pretty close to the observed value of around 6 K (or 6°C). The reason for the small difference could be the limited availability of accurate Mars temperature data, or the uncertainty in the value of the CO2 absorption cross-section, σs.
We know how much radiation from the Sun is arriving at Mars to high accuracy, but knowing how much is being absorbed by the planet surface is more difficult as this depends on an accurate measurement of the Bond albedo. However, conventional astronomical telescopes should be able to measure the reflected radiation to pretty good accuracy as well. That allows us to estimate the expected mean surface temperature without the GHE to fairly high precision. The problem is knowing what the actual surface temperature is with the GHE in operation. This is a difficult enough calculation to do on Earth where we have over 16,000 weather stations measuring the surface temperature on a daily basis, and numerous satellites in orbit. Sadly, none, or very little, of this exists for Mars.
So far in this blog post I have assumed a value of 215 K for the mean surface temperature of Mars, but some reports have put it as high as 225 K (or as low as 210 K). In which case Z = 194.15 and Jth = 37.0. This leads to a 15 µm band stretching from 13.76 µm to 16.47 µm, and a feedback factor of f = 13.0%. Under these circumstances the warming from the Greenhouse Effect increases slightly, but only to 7.7°C.
A Comparison with Earth
For comparison, it is instructive to hypothecate the extent of warming on Earth if its atmosphere also contained 3690 mol/m2 of CO2. In that case Z = 249.3 and Jth = 41.6, which leads to a 15 µm band stretching from 13.62 µm to 16.67 µm, and a feedback factor of f = 14.1%. The resulting predicted temperature rise due to CO2 would be 10.74 K, which is 3.26°C less than the 7.48°C rise currently predicted for Earth as was shown in Post 87). So a twenty-five fold increase in the CO2 concentration would only result in a 3.26°C temperature increase, although as I showed in Post 87, masking by water vapour would probably reduce this by 75% to only 0.8°C.
It is a point of note that the density of CO2 molecules on Mars (3690 mol/m2) is more than double the combined density of all the greenhouse gases on Earth (1560 mol/m2), yet it results in a temperature rise of 5°C - 7°C that is almost ten times less than the 58°C observed for Earth. This is mainly because most of the GHE on Earth is due to water vapour as the width of the CO2 absorption band is so much less than that for water vapour. Even increasing the concentration of CO2 on Mars by a factor of twenty-five cannot appreciably change this.
Summary and Conclusions
What I hope I have shown in this post is that Mars is a good test bed for studying the Greenhouse Effect (GHE). Knowing only its albedo, the atmospheric concentration of CO2, and the intensity of radiation arriving from the Sun, it is possible to accurately predict the temperature rise due to the Greenhouse Effect. This I have predicted to be about 7°C, in close agreement with the current estimate based on observational data (6°C). And this is despite the significant uncertainty over the true measured value of the mean surface temperature on Mars.
The reduced GHE on Mars relative to the Earth occurs despite its much higher (i.e. 24 times greater) atmospheric CO2 concentration. This in turn suggests that the increasing levels of atmospheric CO2 we are currently seeing on Earth will produce only slight temperature increases in the future.
Yes, Mars has its own complicating factors. Heat retention on Mars is limited by the thin atmospheric blanket compared to Earth. This means that the planet does not retain heat very well, but conversely it means that the atmosphere will warm quickly when heated by the Sun. For this reason it may be better to consider Mars under direct solar illumination in daytime. Under these conditions the peak solar flux at the surface of Mars will be four times greater than stated above, or 432 W/m2. This will equate to a peak surface temperature of 295 K (or +22°C). Yet the actual maximum temperature is reported to be around 303 K to 308 K (or +30°C to +35°C). So on this measure the warming from the Greenhouse Effect in daytime near the equator appears to be in the range 8°C to 13°C. Yet the predicted value based on a calculation of the width of the 15 µm absorption band is found to be 10.9°C, in other words in the mid-range of the observed values. Once again this is still much less than the total warming seen on Earth and comparable to the contribution to Earth's GHE seen just from CO2.